"Introduction
The 'Golden Rule of pregnancy' has remained unchanged throughout millennia: Novel and/or potentially harmful substances are never used when new human life is being formed and nurtured within the womb...
Novel and untested medical interventions introduced during any stage of pregnancy are avoided, due to their high potential to cause short- and long-term multigenerational harms that may not be discoverable for years or even decades...
The purpose of Part I presented here is to assess the effects of COVID-19 vaccines on pregnancy outcomes through the Vaccine Adverse Event Reporting System (VAERS) database. VAERS is a national early warning system for vaccine safety established in 1990 as an outgrowth of the National Childhood Vaccine Injury Act signed into law in 1986. VAERS is co-managed by the U.S. Centers for Disease Control and Prevention (CDC) and the U.S. Food and Drug Administration (FDA).
Methods
... The study period was from January 1, 1990 through April 26, 2024, and this includes 412 months for all vaccines except for COVID-19 vaccines that were used only 40 months...
Discussion
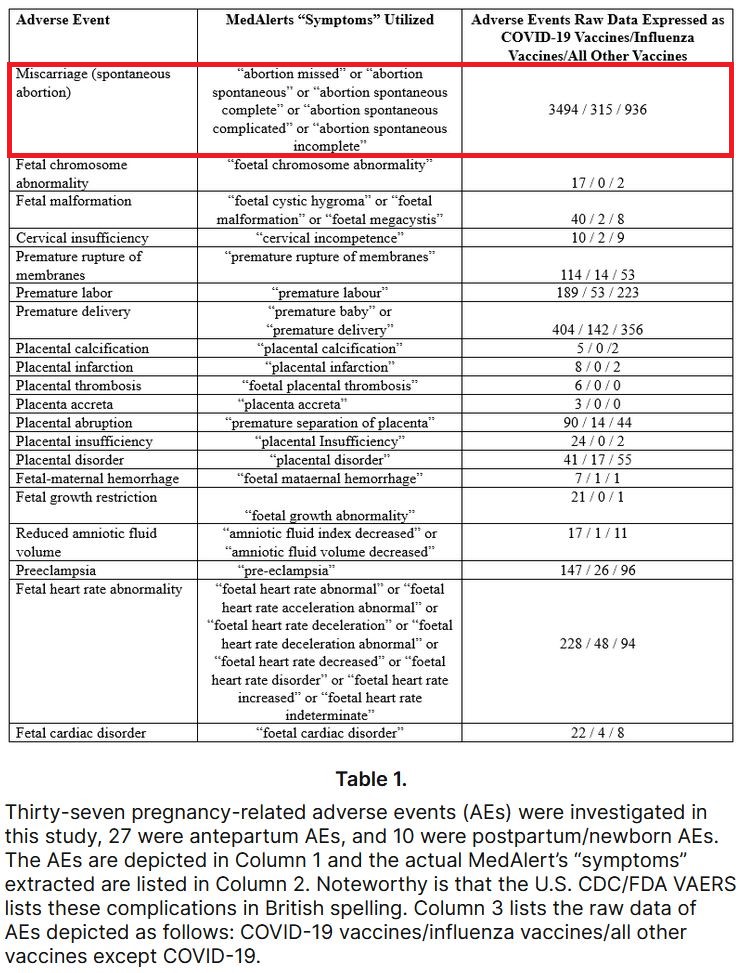
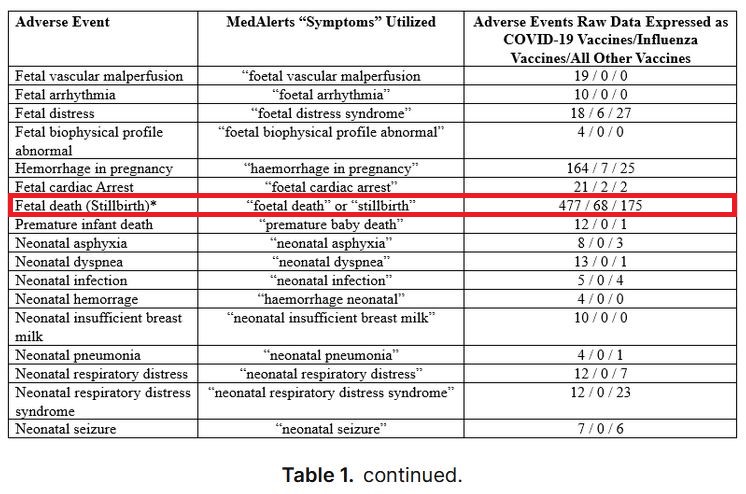
Compared to influenza and all other vaccine products, COVID-19 vaccines in pregnancy have demonstrated unacceptable breaches in safety signals across all 37 AEs investigated, 27 antepartum and 10 postpartum/newborn. All 37 AEs breach CDC/FDA limits for safety and are consistent with the authors’ extensive clinical observations... The magnitude of these safety deviations is unparalleled given the CDC/FDA guidelines...
Conclusion
... The scope of the pregnancy complications in this study are of great concern and consistent with the vast obstetrical experience of the authors’ observations. The catastrophic effects of the COVID-19 vaccines in pregnancy are associated with nearly every obstetrical/neonatal complication imaginable. Placental abnormalities also exhibited a substantial breach in safety signals... The placental abnormalities noted in this study are consistent with clinical observations from sonographers and physicians reviewing ultrasound images prior to birth...
Are Any of Pfizer’s “Randomized, Placebo-Controlled, Double Blinded Clinicals Involving COVID-19 Vaccines Valid?
... As of this report there [are] at least 6 studies demonstrating unequivocal breaches of safety signals with the COVID-19 vaccines in pregnancy. Of these six studies (including this report), three are from the CDC/FDA/VAERS and two from Pfizer... Noteworthy is a recent report documenting that the pharmaceutical industry paid $1.06 billion to reviewers at top medical journals including NEJM, JAMA and BMJ between 2020 and 2022."
© The authors 2025
Terms and Conditions
https://publichealthpolicyjournal.com/terms-and-conditions/
As an open-access journal, we ensure that all published articles are freely available to the global community, enhancing the visibility and impact of authors’ work. Options for copyright transfer by IPAK-EDU, IPAK PHP, and Science Public Health Policy & the Law are not available.
Manuscripts are published under the CC BY 4.0 DEED Attribution 4.0 International https://creativecommons.org/licenses/by/4.0/ by the author.
