¨Abstract
Mass vaccination against coronavirus disease 2019 (COVID-19) is ongoing in many countries worldwide. This study reports the occurrence of acute adverse events among vaccine recipients at a mass vaccination center in Japan. Between August and November 2021, approximately 130,000 individuals received two mRNA vaccine doses (mRNA-1273; Moderna) at the vaccination center. Acute adverse events at the site were observed in 1.1% of the recipients after the first dose and in 0.4% of the recipients after the second dose. The most common event was vasovagal syncope/presyncope, followed by acute allergic reactions...
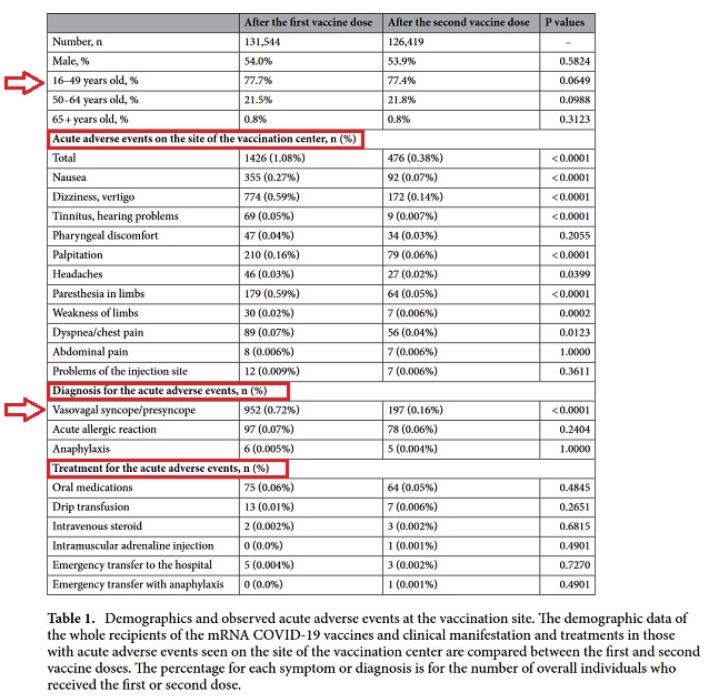
Results
Overall occurrence of acute adverse events on site. A total of 131,544 individuals aged ≥ 16 years were vaccinated with the first dose of the mRNA COVID-19 vaccine at the vaccination center during the study period. Among them, 126,419 (96.1%) were vaccinated for the second dose at the same center 4 weeks after the first dose. The demographics of the enrolled recipients and observed acute adverse events at the vaccination site are summarized in Table 1. Vasovagal syncope/presyncope was the most common acute event for both doses, followed by acute allergic reactions. The occurrence rate of vasovagal syncope/presyncope was much higher after the first dose than after the second dose (0.72% vs. 0.16%, p < 0.0001), but the rate of acute allergic reactions did not differ between the doses (0.07% vs. 0.06%, p = 0.2404)."
¨What is presyncope? Presyncope is staying conscious while feeling like you’re about to faint. Some providers call this common condition near syncope. Syncope is a medical term for fainting.
Presyncope (pree-sing-kuh-pee) can last a few seconds to a few minutes. For most people, it doesn’t cause major problems. But for a small percentage of people who have it, what happens next is more serious. Some people faint after experiencing presyncope and may get hurt when they fall. Others have issues from the cause of their presyncope, like a heart issue.¨
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4. 0/.
